THEORETICAL DEPENDENCE OF
PHOSPHATE POTENTIAL ON
MAGNITUDE OF PROTONMOTIVE
FORCE FOR TWO-ELECTRON
PROTON-CHEMICAL COUPLING
POINT MODEL.
A.G. Anishkin, V.V. Lemeshko.
Biology Research Institute of Kharkov
StateUniversity. 310077, Kharkov, 4 Svobody Sq.
Received September 23, 1998.
At the present t ime the overwhelming majority of works
concerned with the study of energy coupling in biomembranes use
Mitchell's chemiosmotic hypothesis [1] as a
theoretical basis. Nevertheless, some experimentally measured
characteristics of energy coupling system cannot be described in
terms of chemiosmosis [2, 3].
The proton-chemical hypothesis of energy coupling gives possible
variants of the explanation of the mentioned experimental data [4 - 7].
Among the most substantial objections to the orthodox
chemiosmotic coupling principle is the variable ratio of
phosphate potential to the magnitude of protonmotive force [8, 9]. This ratio grows with the
reduction of membrane potential in the process of a titration of
mitochondria in respiration state 4 (by Chance) by uncoupler-protonophore.
The purpose of the present work is the analysis of the
theoretical dependence of the ratio of the phosphate potential to
the membrane potential resulting from the proton-chemical
mechanism of ATP synthesis. This dependence is considered on the
example of a two-electron proton-chemical coupling point. For
this purpose the corresponding electrical model of the system of
mitochondrial oxidative phosphorylation is used.
MODEL
In fig.1 the general system of reactions
in the considered model of oxidative phosphorylation system is
shown. Since the detailed description of a two-electronic proton-chemical
coupling point and the principles of composing of the equivalent
electrical circuits was published earlier [6, 7], it seems expedient
only to mention briefly some key moments of the proton-chemical
point organization. The coupling point is divided in two subunits
"a" and "b" according to [5,
6]. In the subunit "а" the
transfer of the electron pair (2e-) is coupled
only with pumping out 2H+ from a mitochondrion, and in
the subunit "b" 2e- can pass either by
Mitchell's way coupled with pumping out 2H+, or by
chemical way, coupled with synthesis 1 ATP and transfer of 2H+
into mitochondrial matrix.
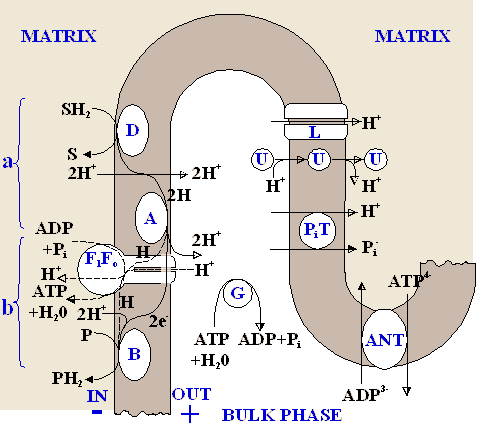 |
Fig.
1. The system of reactions of oxidative
phosphorylation in a two-electronic proton-chemical
coupling point. Symbols: D, A, B, -
redox-centers of a respiratory chain; SH2 and
P - respectively, donor and acceptor of hydrogen atoms; 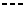 - flow of
electrons through the ATP-synthase (F1Fo)
by the chemical way; ANT adeninnucleotidetranslocase; G -
ferments hydrolysing ATP; PiT - phosphate
carrier; U - uncoupler; L - natural leakage of protons. - flow of
electrons through the ATP-synthase (F1Fo)
by the chemical way; ANT adeninnucleotidetranslocase; G -
ferments hydrolysing ATP; PiT - phosphate
carrier; U - uncoupler; L - natural leakage of protons.
|
The given system of energy coupling can be modelled by the equivalent electrical circuit given in fig. 2. The circuit consists of five contours.
The contour I describes a flow of electrons through the whole
coupling point. The current J corresponds to a flow of electrons,
the voltage Ео reflects the difference of
redox potentials DEBD, ro corresponds to internal resistance of Ео
source. The voltage ea and eb in contour I
and other contours reflects respectively redox potential decrease
DEAD and DEBA in the subunits "a"
and "b" of a proton-chemical point.
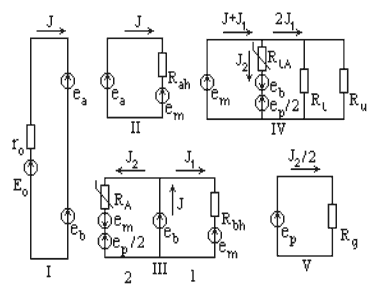 |
Fig. 2. The equivalent electrical
circuit of a two-electron proton-chemical coupling point. See comments in the text.
|
Contour II shows the coupling of electron flow with pumping
out of protons in the subunit "a". The resistance Rah
characterizes the work of the appropriate proton pump. The
voltage em corresponds to the transmembrane potential DmH+.
Contour III describes the coupling of electrons transfer in
the subunit "b" of a respiratory chain with pumping out
of protons (flow J1) and ATP synthesis (flow J2).
In the given contour the resistance Rbh corresponds to
H+-pump of the subunit "b", and the voltage
ep describes the phosphate potential in
nonmitochondrial bulk phase. The resistance RA
reflects the work of the ATP-synthase. According to the redox
nature of the formation of "high-energy" bond in the
proton-chemical mechanism of ATP synthesis [4 - 7], RA exponentially depends on the
magnitude of DmH+ and redox
potential eb. It is reflected in the
equation (6), where RA0 -
normalising multiplier.
Contour IV describes the distribution of proton flows in the
simulated system. In the given contour resistance RL
reflects natural leakage of protons through a membrane, and RU
describes the leakage caused by an added uncoupler. The
resistance RLA describes the leakage of protons
through the ATP-synthase and system of phosphorylation substrates
transport coupled with ATP synthesis.
Contour V displays ATP hydrolysis in energy-consuming
processes determined by the corresponding resistance Rg.
The given equivalent electrical circuit can be described by
the following system of eight equations, in which there are eight
unknown variables.
The choice of numerical values of resistances was based on the
general qualitative characteristics of mitochondrial oxidative
phosphorylation system, such as low proton conductivity of a
coupling membrane, approximate value of the respiratory control
ratio, the maximum degree of respiration activation, and others.
For some parameters, such as a normalising multiplier of the ATP-synthase
resistance, which are not obviously determined by general reasons
the selection was carried out empirically with the subsequent
computer analysis of the model behaviour. (The more detailed
description of the parameters' values choice see in work [6].) The values of resistances given below are
normalized per one coupling point and have a relative character,
though they are expressed in ohms.
The following values of parameters of the electrical circuit
were chosen: Eo = 400 mV; ro = 1 ohm, Rah,
Rbh = 10 ohms; RA0 = 109 ohms, RL
= 100 ohms; Rg. = 1000 ohms. The titration of system
by uncoupler was simulated by the resistance RU change
from value 1000 ohms corresponding to state 4 of respiration (by
Chance), up to value 1 ohm at the maximum concentration of
uncoupler.
RESULTS AND DISCUSSION
The solving of the equations system (1) - (8) was carried out by the computer. The computer
solving of the equations system allowed us to receive the
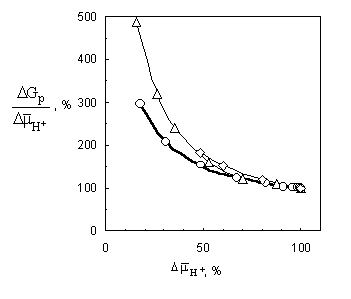
dependences given in fig. 3. As can be seen,
with the decrease of the membrane potential the ratio DGP/ DmH+ grows.
From the point of view of the proton-chemical coupling principle
it can be explained as follows. For ATP synthesis both the energy
and directly the energy of redox potentials difference in a
respiratory chain are used. At the uncoupler addition the part of
energy received from protonmotive force is reduced much more than
the part of energy received from the redox-potentials difference.
Due to this the ratio of the phosphate potential (including both
these parts) to the membrane potential grows.
Thus, the redox status of the redox-centers of a respiratory
chain directly influences the characteristics of ATP formation
system. This is inherent qualitaty of a proton-chemical coupling
principle in contrast to delocalized chemiosmosis. The obtained
curves (fig. 3) qualitatively correlate with
real experimental data [8, 9],
contradicting to the chemiosmotic hypothesis. Also good agreement
with the experimental data, incompatible with the delocalized
chemiosmosis principle, was shown earlier for theoretical
dependences of ATP synthesis and respiration rates on the
membrane potential when the latter was changed in various ways [6, 7]. Thus, the analysis of the
model of the proton-chemical coupling mechanism can be considered
as an argument in favor of a principle possibility of its
existence.
LITERATURE
- Mitchell P.; Nature (London), 1961, v.
191, pp. 144-148
- Ferguson S.J.; Biochem. et Biophys. Acta,
1985, v.811, N1, pp. 47- 95.
- Slater E.C., Berden J.A., Herweijer M.A.;
Biochem. et Biophys. Acta, 1985, v. 811, N 1, pp. 217-231.
- Lemeshko V.V.; Dokl. Akad. Nauk USSR, ser.
B, 1980, N 10, p. 78-81.
- Lemeshko V.V.; Kiev: Nauk. Dumka; Bikh.
Giv. Chel., 1988, N.
12, p. 54- 65.
- Lemeshko V.V., Anishkin A.G.; Biofizika.,
- 1998, - N 2, - p. 308-314
- Anishkin A.G., Dop. Nacion. Akad. Nauk.
Ukr., 1998, N5, p. 183-189
- Petronili V., Pietrobon D., Zoratti M.,
Azzone J.F.; Eur. J. Biochem., 1986, v. 155, N 2, pp. 423-431.
- Westerhoff H.V., Simonetti A.L.M., Van
Dam K.; Biochem. J., 1981, v. 200, pp. 193 - 202.

![]() - flow of
electrons through the ATP-synthase (F1Fo)
by the chemical way; ANT adeninnucleotidetranslocase; G -
ferments hydrolysing ATP; PiT - phosphate
carrier; U - uncoupler; L - natural leakage of protons.
- flow of
electrons through the ATP-synthase (F1Fo)
by the chemical way; ANT adeninnucleotidetranslocase; G -
ferments hydrolysing ATP; PiT - phosphate
carrier; U - uncoupler; L - natural leakage of protons.